The Bohr Model of the Atom
Its 1913, the world of particle physics will change again. Niels Bohr, a Danish physicist and a student of Rutherford, proposes a new model for the Hydrogen atom. Bohr’s model, inspired from Rutherford’s model, retains the defined electron orbitals. More importantly, it’s able to explain the line spectrum for hydrogen because it incorporates several new ideas about energy.
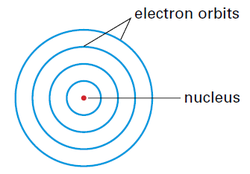
The model of the atom in 1913. Although Bohr pictured the atom in three dimensions, it is presented here, in two dimensions, to simplify some of Bohr’s revolutionary concepts.
The model of the atom in 1913. Although Bohr pictured the atom in three dimensions, it is presented here, in two dimensions, to simplify some of Bohr’s revolutionary concepts.
Bohr’s atomic model pictures electrons in orbit around a central nucleus; however, unlike Rutherford’s model, it places restrictions upon the movement of electrons (specifically, electrons are allowed to move anywhere within the volume of space around the nucleus).
What was Bohr’s basis in making these claims? Simply put, Max Planck.
Max Planck

In 1900, a physicist named Max Planck proposed an idea so radical, that it was unfathomable even to him. It was so revolutionary that he himself was unwilling to accept its implications. Planck suggested that matter, at the atomic level, can absorb or emit only discrete quantities of energy. He called these specific quantities, quanta. In other words, Planck stated that the energy of an atom is quantized, meaning that it can only exist in certain discrete amounts. It is not continuous.
Example
Rungs of a ladder are quantized because they are finite (exist in discrete amounts).
Smooth slope of a ramp is not quantized because it is infinite (exists in infinite amounts).
Unlike ladders or ramps, a quantum is an extremely small “packet” of energy.
Although Planck said that energy of matter is quantized (meaning energy could be dualistic in nature, both as a wave and as particles), he was unwilling to admit that energy might have particle-like properties.
In 1905, Albert Einstein made this assertion. Light, according to Einstein, is also quantized. It occurs as quanta of EM energy made up of particle-like “packets” of energy called photons. Einstein hypothesized that light (and by extension, all EM energy) travels in the form of photons or energy. Therefore, light is emitted as photons of energy, and light is absorbed as photons of energy.
Example
Rungs of a ladder are quantized because they are finite (exist in discrete amounts).
Smooth slope of a ramp is not quantized because it is infinite (exists in infinite amounts).
Unlike ladders or ramps, a quantum is an extremely small “packet” of energy.
Although Planck said that energy of matter is quantized (meaning energy could be dualistic in nature, both as a wave and as particles), he was unwilling to admit that energy might have particle-like properties.
In 1905, Albert Einstein made this assertion. Light, according to Einstein, is also quantized. It occurs as quanta of EM energy made up of particle-like “packets” of energy called photons. Einstein hypothesized that light (and by extension, all EM energy) travels in the form of photons or energy. Therefore, light is emitted as photons of energy, and light is absorbed as photons of energy.


 RSS Feed
RSS Feed
