Electrodeposition
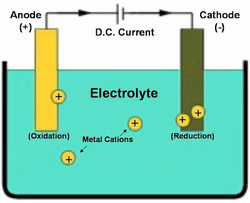
Chemistry, especially at higher levels, requires having a working understanding of basic chemical processes and concepts. Having a basic understanding of how electro-deposition works in necessary prior to conducting any electroplating experiments.
Electrodeposition McMaster University
Electrodeposition McMaster University
Electroplating Copper onto Nickel
Most pure metals don’t have the desired physical properties or corrosion resistance necessary for the many applications of metals in our society. The most beautiful metals, such as Silver (Ag) and Gold (Au), can also be the most expensive, and in their purest forms, the most fragile. Pure gold is soft and pure silver tarnishes very quickly.
Electroplating is a common method used to produce beautiful and durable metal objects. Jewelry and other metal objects can be made cheaper, stronger, and more durable by coating a strong, cheaper, metal with a thin layer of a more expensive metal.
For example, cheaper jewelry can be made from copper or nickel, and then coated with silver. More expensive jewelry can be made from silver, and then coated with gold. Industry also uses electroplating for many applications. Chromium, an extremely hard surface metal, is commonly electroplated over steel cores for heavy duty applications. Worn or damaged metal machine parts may be restored by re-plating the worn sections.
Electroplating is a common method used to produce beautiful and durable metal objects. Jewelry and other metal objects can be made cheaper, stronger, and more durable by coating a strong, cheaper, metal with a thin layer of a more expensive metal.
For example, cheaper jewelry can be made from copper or nickel, and then coated with silver. More expensive jewelry can be made from silver, and then coated with gold. Industry also uses electroplating for many applications. Chromium, an extremely hard surface metal, is commonly electroplated over steel cores for heavy duty applications. Worn or damaged metal machine parts may be restored by re-plating the worn sections.
| electroplating_lab.pdf |
| electroplating_lab.docx |

 RSS Feed
RSS Feed
