
The history of atomic history, and thus Chemistry as a whole, is full of successes and failures.
It was the Ancient Greek philosopher by the name of Democritus in about 300 BC, who hypothesized that matter reaches a point of divisibility. This means that there is some point where nothing can be divided any further (i.e. basic building block of all matter). The named this ‘building block’ atomos - literally meaning “indivisible”.
Fast-forward about 2000 years.
It’s 1805. The Age of Enlightenment is in full swing. An English school teacher and chemist by the name of John Dalton recreates the modern theory of atoms so that he can explain 3 major scientific laws:
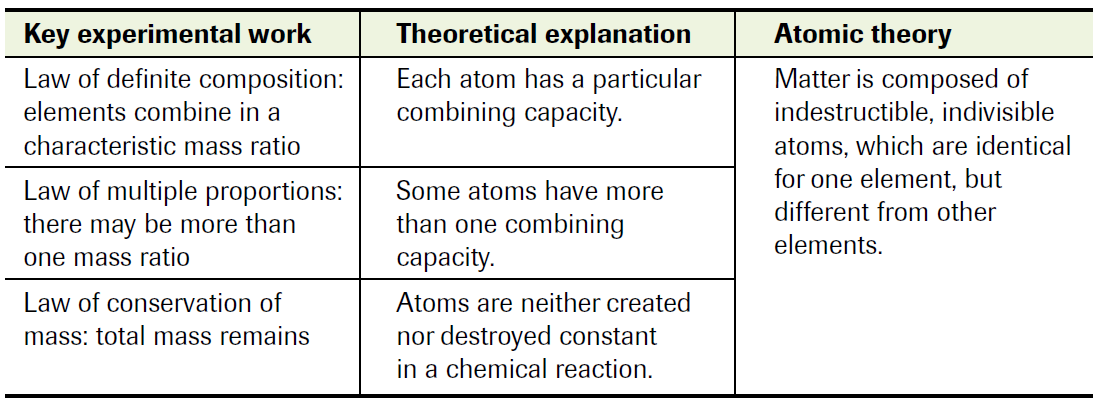
1. Law of Definite Composition
2. Law of Multiple Proportions
3. Law of Conservation of Mass
Dalton’s theory (or is it Democritius’?) successfully explained all 3 of these laws. The primary tenet of his theory was the smallest piece of matter, the atom, which he described as being not only indivisible, but also different from one element to another. The best way to understand is via an analogy, one that Dalton provided himself – a billiard ball (a tiny, solid, indestructible sphere)
It was the Ancient Greek philosopher by the name of Democritus in about 300 BC, who hypothesized that matter reaches a point of divisibility. This means that there is some point where nothing can be divided any further (i.e. basic building block of all matter). The named this ‘building block’ atomos - literally meaning “indivisible”.
Fast-forward about 2000 years.
It’s 1805. The Age of Enlightenment is in full swing. An English school teacher and chemist by the name of John Dalton recreates the modern theory of atoms so that he can explain 3 major scientific laws:
1. Law of Definite Composition
2. Law of Multiple Proportions
3. Law of Conservation of Mass
Dalton’s theory (or is it Democritius’?) successfully explained all 3 of these laws. The primary tenet of his theory was the smallest piece of matter, the atom, which he described as being not only indivisible, but also different from one element to another. The best way to understand is via an analogy, one that Dalton provided himself – a billiard ball (a tiny, solid, indestructible sphere)

 RSS Feed
RSS Feed
