The Nuclear Atomic Model
Toward the close of the 19th century, chemists had 2 invaluable conceptual tools to aid them in their understanding of matter – John Dalton’s Atomic Theory and Dmitri Mendeleev’s Periodic Table.
As you know, neither of the 2 are applicable, as both have undergone significant changes. When Mendeleev invented the periodic table, he was well-acquainted with Dalton’s atomic theory. He knew nothing, however, about subatomic particles, and especially the electron, which is the foundation for the modern periodic table’s distinctive shape. Original periodic table was developed out of experimentation rather than research and understanding.
As you know, neither of the 2 are applicable, as both have undergone significant changes. When Mendeleev invented the periodic table, he was well-acquainted with Dalton’s atomic theory. He knew nothing, however, about subatomic particles, and especially the electron, which is the foundation for the modern periodic table’s distinctive shape. Original periodic table was developed out of experimentation rather than research and understanding.
The Discovery of the Electron Requires a New Atomic Model
For the most of the 19th century, scientists toyed with electricity, led by Svante Arrhenius and Michael Faraday. Studies by these two, combined with research done by William Crookes with electricity and vacuum tubles, suggested that electric charges were compounds of matter.
Thomson’s Atomic Model
 The atomic model in 1903. Thomson viewed the atom as a positively charged sphere embedded with sufficient numbers of electrons to balance (neutralize) the total charge.
The atomic model in 1903. Thomson viewed the atom as a positively charged sphere embedded with sufficient numbers of electrons to balance (neutralize) the total charge. The year of 1897 starts off with a bang. Dalton’s idea of an indivisible atom is shattered. Another Englishman, J.J Thomson discovers the existence of a negatively charged particle, the electron. It takes a few years for chemists to really understand the consequences and ramifications of this discovery. Someone finally puts 2 and 2 together, and realizes that if atoms contain negatively charged particles, they must also contain positively-charged particles (to balance things out).
The atomic model that Thomson proposed is shown below. Keep in mind that scientists haven’t yet discovered the proton. Therefore, in Thomson’s model, the entire sphere carries a uniform, positive charge.
The atomic model that Thomson proposed is shown below. Keep in mind that scientists haven’t yet discovered the proton. Therefore, in Thomson’s model, the entire sphere carries a uniform, positive charge.

Another way to imagine this is using a raisin bun as an example. Imagine the raisins as the electrons and the bun (the bread) as the positive “area”.
Rutherford’s Nuclear Model of the Atom
Okay, so before we get to Rutherford and his model, we have to understand the scientific discoveries that made his research/model possible.
In the last 4 years of the 19th century, scientists in France – notably Henri Becquerel and Pierre and Marie Curie – discovered that certain elements are radioactive. That is, their atoms naturally emit positively charged particles (α-particles), negatively charged particles (β-particles), and energy (γ-radiation).
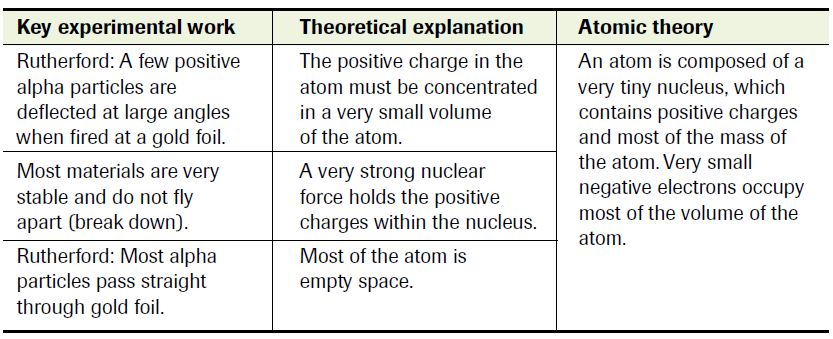
So let’s jump 10 years. Its 1908, and Ernest Rutherford, another English chemist (see a trend?), just wins a Nobel Prize in Chemistry “for his investigations into the disintegration of the elements, and the chemistry of radioactive substances”. One year later, 2 of Rutherford’s students report observations that cast doubts on Thomson’s atomic model. Rutherford and his students, as a part of their investigation into the nature and properties of radioactive emissions, aim α-particles at extremely thin metal foils. They observe that about 1 in every 8000 α-particles are deflected significantly by the metal foils’ atoms. The numbers disagree with Thomson’s atomic model (and thus his conclusions). Researchers expect them -particles to pass through the metal atoms with deflections averaging 1/200 of a degree. What they observe are deflections of 90 degrees and more, straining the credibility of Thomson’s model.
Rutherford concludes that either his observations/data are flawed, or Thomson’s model is. In 1911, Rutherford conducts the now-famous gold-foil experiment.
In the last 4 years of the 19th century, scientists in France – notably Henri Becquerel and Pierre and Marie Curie – discovered that certain elements are radioactive. That is, their atoms naturally emit positively charged particles (α-particles), negatively charged particles (β-particles), and energy (γ-radiation).
So let’s jump 10 years. Its 1908, and Ernest Rutherford, another English chemist (see a trend?), just wins a Nobel Prize in Chemistry “for his investigations into the disintegration of the elements, and the chemistry of radioactive substances”. One year later, 2 of Rutherford’s students report observations that cast doubts on Thomson’s atomic model. Rutherford and his students, as a part of their investigation into the nature and properties of radioactive emissions, aim α-particles at extremely thin metal foils. They observe that about 1 in every 8000 α-particles are deflected significantly by the metal foils’ atoms. The numbers disagree with Thomson’s atomic model (and thus his conclusions). Researchers expect them -particles to pass through the metal atoms with deflections averaging 1/200 of a degree. What they observe are deflections of 90 degrees and more, straining the credibility of Thomson’s model.
Rutherford concludes that either his observations/data are flawed, or Thomson’s model is. In 1911, Rutherford conducts the now-famous gold-foil experiment.
 Rutherford's Nuclear Model of the Atom
Rutherford's Nuclear Model of the Atom On the basis of his experiment, Rutherford suggests that the deflections observed are caused by an encounter between positively-charged α-particles and an intense electric field at the centre of the atom.
After doing the math, Rutherford arrives at an inescapable conclusion: the atom is made up mainly of empty space, with a massive region of concentrated charge at the centre. A few years later, the charge is determined to be positive, and is named the atomic nucleus.
Rutherford designs his own atomic model, now known as the nuclear model of the atom, where electrons travel around the nucleus in defined paths (think planets revolving around the Sun).
After doing the math, Rutherford arrives at an inescapable conclusion: the atom is made up mainly of empty space, with a massive region of concentrated charge at the centre. A few years later, the charge is determined to be positive, and is named the atomic nucleus.
Rutherford designs his own atomic model, now known as the nuclear model of the atom, where electrons travel around the nucleus in defined paths (think planets revolving around the Sun).



 RSS Feed
RSS Feed
