Prelude to a New Atomic Model
Rutherford’s atomic model have solved the inherent problems in Thomson’s atomic model, but it also raised others:
- An atomic nucleus composed entirely of positive charges should fly apart due to electrostatic forces of repulsion
- Rutherford’s nuclear atom couldn't adequately explain the total mass of the atom
Atomic Spectra
Electromagnetic Energy is energy carried through space or matter by means of wavelike oscillations. Think waves (something goes up and down). The oscillations are systematic fluctuations in the intensities of very tiny electrical and magnetic forces. The space between these oscillations is called the Electromagnetic Field. Each oscillations is called 1 cycle. Successive series of these oscillations is called Electromagnetic Radiation (in other words, light wave).
The unit of measuring frequency is called, in SI terms, hertz (Hz)
As the radiation moves away from its source, the maximum values of the EM forces are regularly spaced. The distance separating maximum values is called the wavelength (λ), measured in meters (m).
If we multiply the wavelength (λ) by frequency (f ), the result is the velocity.
If we multiply the wavelength (λ) by frequency (f ), the result is the velocity.
EM radiation comes in large range of frequencies called the Electromagnetic Spectrum. Remember that there is an inverse relationship between wavelength and frequency.
The visible portion of the EM spectrum is called the Continuous Spectrum, because the component colors are indistinct. They appear “smeared” together into a continuum of color. When atoms absorb energy (ex. When they are exposed to an electric current), a pattern of discrete (distinct) colored lines separated by spaces of varying lengths is observed.






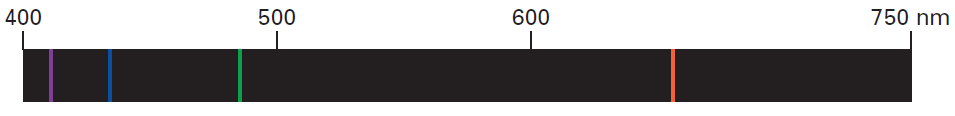
 RSS Feed
RSS Feed
