The Quantum Mechanical Model
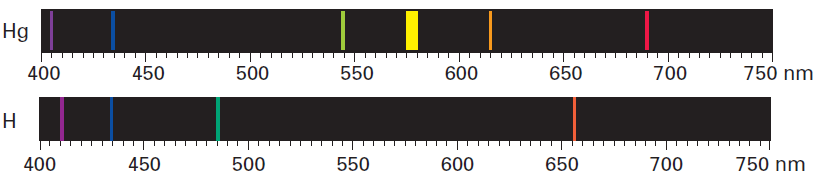
Bohr’s model ma have explained the emission spectrum of atoms with only 1 electron; however, it failed to explain the emission spectra for multi-electron atoms, like Mercury.
Because Mercury has more atoms than a Hydrogen atom, it also has more spectral lines. Observations like these forced Bohr and other scientists to reconsider the nature of energy levels. The large spaces between the individual colors suggested that there are energy differences between individual energy levels. The smaller spaces between color lines, however, suggested that there were smaller energy differences within energy levels.
In other words, scientists hypothesized that there are sublevels within each energy level.
Even after modifying Bohr’s model to include the idea of energy sublevels, the fundamental problem with Bohr’s model still wasn’t resolved. The model still couldn’t explain multi-electron atoms.
The solution to this problem wasn’t possible until another important property of matter was discovered.
In other words, scientists hypothesized that there are sublevels within each energy level.
Even after modifying Bohr’s model to include the idea of energy sublevels, the fundamental problem with Bohr’s model still wasn’t resolved. The model still couldn’t explain multi-electron atoms.
The solution to this problem wasn’t possible until another important property of matter was discovered.
The Discovery of Matter Waves
Energy’s matter-like properties became standard knowledge by the early 1920s. In 1924, a physicist by the name of Louis de Broglie hypothesized a concept that would eventually become a cornerstone for the modern Quantum model. de Broglie asked: “What if matter has wave-like properties?”
He developed an equation that enabled him to calculate the wavelength associated with any object – large, small, or microscopic.
Example
A baseball with a mass of 142g and moving with a speed of 25.0m/s has a wavelength of 2 × 10-34 m.
Objects we can see and interact with, like baseballs, have wavelengths so small that they don’t have any significant observable effect on the object’s motion. However, for microscopic objects like electrons, the effect of wavelength on motion becomes very significant.
Example
An electron moving at a speed of 5.9 × 10^6 m/s has a wavelength of 1 × 10^10 m. the size of this wavelength is greater than the size of the hydrogen atom (radius = 5.3 × 10-11 m) to which it belongs.
It wasn’t until 1927 when de Broglie’s hypothesis received experimental support. Researchers observed that streams of moving electrons produced diffraction patterns similar to those that are produced by waves of EM radiation. Since diffraction involves the transmission of waves through a material, the observation supported the idea that electrons had wave-like properties.
He developed an equation that enabled him to calculate the wavelength associated with any object – large, small, or microscopic.
Example
A baseball with a mass of 142g and moving with a speed of 25.0m/s has a wavelength of 2 × 10-34 m.
Objects we can see and interact with, like baseballs, have wavelengths so small that they don’t have any significant observable effect on the object’s motion. However, for microscopic objects like electrons, the effect of wavelength on motion becomes very significant.
Example
An electron moving at a speed of 5.9 × 10^6 m/s has a wavelength of 1 × 10^10 m. the size of this wavelength is greater than the size of the hydrogen atom (radius = 5.3 × 10-11 m) to which it belongs.
It wasn’t until 1927 when de Broglie’s hypothesis received experimental support. Researchers observed that streams of moving electrons produced diffraction patterns similar to those that are produced by waves of EM radiation. Since diffraction involves the transmission of waves through a material, the observation supported the idea that electrons had wave-like properties.
The Quantum Mechanical Model of the Atom
In 1926, an Austrian physicist, Erwin Schrodinger, used math and statistics to combine de Broglie’s idea of matter waves and Einstein’s idea of quantized energy particles (photons). Schrodinger’s mathematical equations and their interpretations, together with another idea called Heisenberg’s uncertainty principle, resulted in the birth of the field of quantum mechanics (a branch of physics that uses mathematical equations to describe the wave properties of sub-microscopic particles such as electrons, atoms, and molecules). Schrodinger used concepts from quantum mechanics to propose a new atomic model: the quantum mechanical model of the atom. This model describes atoms as having certain allowed quantities of energy because wave-like properties of their electrons.
The image above depicts the volume surrounding the nucleus of the atom as being indistinct or cloud-like because of a scientific principle called the uncertainty principle. The uncertainty principle was proposed in 1927 by a German physicist named Werner Heisenberg, showing that it is impossible to known both the position and the momentum of an object beyond a certain measure of precision (an object’s momentum is a property given my its mass multiplied by its velocity).
According to the principle, if you can know an electron’s precise position and path around the nucleus, as you would by defining its orbit, you cannot know with certainty its velocity. Similarly, if you know its precise velocity, you cannot know with certainty its position. Based on the uncertainty principle, Bohr’s atomic model is flawed because you cannot assign fixed paths (orbits) to the motion of electrons.
Clearly, however, electrons exist. And if they exist, they must exist somewhere. To describe where that “somewhere” is, researchers use an idea from a branch of math called statistics. Although you can’t talk about electrons in terms of certainties, you can talk about them in terms of probabilities. Schrodinger used a type of equation called a wave equation to define the probability of finding an atom’s electrons at a particular point within the atom. There are many solutions to this wave equation and each solution represents a particular wave function, which gives information about an electron’s energy and location within an atom. These wave functions are called orbitals.
According to the principle, if you can know an electron’s precise position and path around the nucleus, as you would by defining its orbit, you cannot know with certainty its velocity. Similarly, if you know its precise velocity, you cannot know with certainty its position. Based on the uncertainty principle, Bohr’s atomic model is flawed because you cannot assign fixed paths (orbits) to the motion of electrons.
Clearly, however, electrons exist. And if they exist, they must exist somewhere. To describe where that “somewhere” is, researchers use an idea from a branch of math called statistics. Although you can’t talk about electrons in terms of certainties, you can talk about them in terms of probabilities. Schrodinger used a type of equation called a wave equation to define the probability of finding an atom’s electrons at a particular point within the atom. There are many solutions to this wave equation and each solution represents a particular wave function, which gives information about an electron’s energy and location within an atom. These wave functions are called orbitals.


 RSS Feed
RSS Feed
